Successful Development of an artificial intelligence (a deep learning) model to classify pancreatic adenocarcinoma in endoscopic ultrasound-guided fine needle aspiration (EUS-FNA) biopsy histopathological specimens. ― Published in Scientific Reports ―
Medmain Inc. (Headquarters: Fukuoka City, Fukuoka Prefecture, CEO: Osamu Iizuka, hereinafter “Medmain”), a provider of “PidPort” pathological diagnosis support solutions, has succeeded in developing an artificial intelligence model to classify pancreatic adenocarcinoma in endoscopic ultrasound-guided fine needle aspiration (EUS-FNA) biopsy histopathological specimens through a multi-institutional joint research with Kurume University Hospital and other medical institutions nationwide. With this development, we are now able to detect pancreatic adenocarcinoma among pancreatic cancers. A paper on this development was published on April 19, 2021 in “Scientific Reports”, a natural science journal issued by Nature Research in the United Kingdom.

■Outline of the Research Results
We have established an artificial intelligence (a deep learning) model to discriminate pancreatic adenocarcinoma in endoscopic ultrasound-guided fine needle aspiration (EUS-FNA) biopsy histopathological specimens.
■Background of the Results
The pancreatic endoscopic ultrasound-guided fine needle aspiration (EUS-FNA) biopsy is a method to observe submucosal and/or extramural lesions or tumors in the thorax, abdomen, or pelvis with an ultrasound endoscope from gastrointestinal tracts (e.g., stomach or duodenum), and collect cells by puncture with a fine-needle. In Japan, both morbidity and mortality rates of pancreatic cancer are increasing, and the lesion may already have reached an advanced stage by the time the patient shows symptoms. Therefore, the pancreas is called the “silent organ”.
To determine a treatment strategy, cytopathological diagnosis is crucial along with accurate medical image (e.g., radiological) diagnosis. The pancreatic EUS-FNA method has been widely used mainly in European countries and the United States. In Japan, since it has been covered by health insurance in 2010, it has started to be used widely as well and is now becoming a mainstream method for the diagnosis of pancreatic cancers. However, because cell and tissue fragments collected by the EUS-FNA method are very small, pathological diagnosis is difficult in such cases.
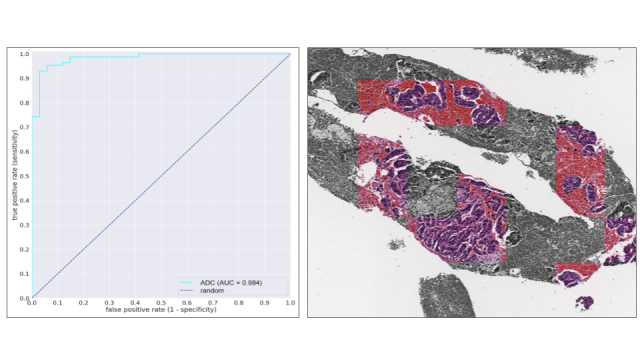
The aim of this study is to develop an artificial intelligence model using deep learning approach that can detect pancreatic adenocarcinoma in EUS-FNA biopsy histopathological specimen digital images.
■Methods
In this study, we have received pancreatic EUS-FNA biopsy specimens from Kurume University Hospital, and after digitizing, a number of collaborative pathologists in Japan created the training sets and developed the artificial intelligence model by deep learning. The accuracy of the developed model was verified by using a test set consisting of cases with consensus diagnosis was obtained by three pathologists specializing in pancreatic pathology.
■Results
In the present study, we have achieved very high accuracy (ROC-AUC, 0.98; accuracy rate, 94%; sensitivity, 0.93; and specificity, 0.97) to discriminate pancreatic adenocarcinomas. Moreover, the predicted histopathological areas of adenocarcinoma by our deep learning model were confirmed to be appropriate by pathologists. In conclusion, we have succeeded in developing an artificial intelligence model that can discriminate pancreatic adenocarcinoma with high accuracy in EUS-FNA biopsy specimens.
Following our published papers since last year (2020) on the development of artificial intelligence models for the stomach, colon, and lungs, we are now able to discriminate pancreatic adenocarcinoma in small biopsy specimens.

We are planning to verify the accuracy of the existing artificial intelligence model at a number of facilities in the next step.
■Original Article
The results of this research were published in the online edition of Scientific Reports (April 14, 2021).
▼Paper Title: A deep learning model to detect pancreatic ductal adenocarcinoma on endoscopic ultrasound-guided fine-needle biopsy
Scientific Reports
▼Japanese Translation: 超音波内視鏡下穿刺吸引生検病理組織デジタル標本における膵癌の検出を可能にする深層学習を用いた人工知能の開発
▼DOI:https://www.nature.com/articles/s41598-021-87748-0
■Author information
<Department of Pathology, Kurume University School of Medicine>
Hirohisa Yano, Yoshiki Naito
<Department of Diagnostic Pathology, Kurume University Hospital>
Jun Akiba
<Department of Pathology, Jichi Medical University>
Noriyoshi Fukushima
<Department of Anatomic Pathology, Graduate School of Medical Sciences, Kyushu University>
Yoshinao Oda, Yuichi Yamada
<Department of Diagnostic Pathology, Kyushu Cancer Center>
Yutaka Koga
<Department of Pathology, Kagoshima University Graduate School of Medical and Dental Sciences>
Michiyo Higashi, Hirotsugu Noguchi
<Department of Pathology, Kurashiki Central Hospital>
Kenji Notohara
Kaori Uchino, Junya Itakura
<Department of Pathology and Microbiology, Saga Medical School Faculty of Medicine, Saga University>
Shinichi Aishima
<Department of Pathology, Department of Pathology, Saga University Hospital>
Keita Kai
<Department of Pathology, Shizuoka Cancer Center>
Nobuyuki Ohike
Tomoko Norose
<Department of Diagnostic Pathology, Tokai University Hachioji Hospital>
Takuma Tajiri
<Department of Pathology, Saitama Medical University>
Hiroshi Yamaguchi
<Department of Human Pathology, School of Medicine, Juntendo University>
Yuki Fukumura
<Division of Pathology, Exploratory Oncology Research and Clinical Trial Center, National Cancer Center>
Motohiro Kojima
<Department of Pathology, Tokai University School of Medicine>
Kenichi Hirabayashi
<Department of Pathology, Faculty of Medicine, Fukuoka University>
Yoshihiro Hamada
<Department of Investigative Pathology, Tohoku University Graduate School of Medicine>
Toru Furukawa, Yuko Omori
<Department of Anatomic Pathology, Tokyo Medical University>
Aoi Sukeda
<Division of Gastroenterology, Department of Medicine, Kurume University School of Medicine>
Yoshinobu Okabe
<Medmain Inc.>
Masayuki Tsuneki, Fahdi Kanavati
■Company Overview
【Company Name】Medmain Inc.
*Ministry of Economy, Trade and Industry J-START UP, Selected Company https://www.j-startup.go.jp/startups/
【Date of Establishment】01/11/2018
【Profile】Medical Software, Planning・Development・Operation and Sales of Cloud Services
【CEO】Osamu Iizuka
【Address】【Tokyo Office】2-10-11 Minami Aoyama #A Aoyama Bldg. 2F, Minato-ku, Tokyo 【Fukuoka Office】2-4-5 Akasaka #104, Chuo-ku, Fukuoka
■Related Sites
【Official Corporate Website】 https://medmain.com/
【Pathology Diagnosis Support Solution「PidPort」】 https://pidport.medmain.com/
【Imaging Center | Digitization Services for Pathology Specimens】 https://imaging.medmain.com/
■Contact us
Medmain Inc.
Sato (Mr.) at PR Dept. : pr-m@medmain.com